Breast Cancer Prognosis: A Genetic Code for Personalized Therapy
Introduction
Today, breast cancer is distinguished into at least three different subtypes based on clinical and molecular parameters: luminal, erbB2, and basal type, which exhibit different biological behaviors and prognoses. Correctly identifying the molecular subtype of the tumor opens the door to new, increasingly adequate and targeted therapeutic possibilities for the treatment of the specific molecular subtype [1]. In this scenario, luminal carcinomas (which represent approximately 65% of the total) are distinguished by a particular heterogeneity of biological behavior, with recovery of disease in approximately 40-50% and death in approximately two-thirds of these patients 5 years after diagnosis, despite initial anatomopathological pictures of apparent low aggressiveness [2- 9]. Precisely for this diagnostic category, biomolecular parameters derived from the genome/transcriptome that are capable of orienting the therapeutic choices in a more precise and personalized way on the patient’s actual therapeutic needs are desirable [10,11]. Currently, the clinical (T, nodal status) and biopathological (hormonal status) parameters obtained from membrane receptor expression provide prognostic information and an indication of any adjuvant systemic chemoradiotherapy treatment [12-14]. However, adjuvant therapy reduces the risk of recurrence by only 25-30% [13]. These data are probably due to:
1. Clinicopathological parameters of stratification of the risk of disease recovery are not sufficiently adequate for the patient’s prognostic framework.
2. Adjuvant therapies are not specific enough toward the cells responsible for disease recovery [15,16].
Knowing the details of the mutations of every single tumor allows us to predict the biological behavior of that neoplasm and to adequately stratify the risk. Genetic tests, such as Mammaprint and Oncotype DX, EndoPredict [17,18], assist clinicians in choosing the most suitable adjuvant treatment by analyzing the expression profile of genes involved in the metastasis process (St Gallen 2017). These tests are very expensive and often have to be sent to foreign laboratories. The genetic profile is of the utmost importance in the evaluation of parameters already known as the expression of hormonal receptors and HER2; these are currently determined with immunohistochemistry (IHC) or FISH methods, which provide information about the morphological expression of the receptor but not about their functional state. In any case, knowing that the receptor is expressed at the membrane level is not sufficient information to guarantee the effectiveness of the drug addressed to it because that protein may not be functionally active. Therefore, it is necessary to ascertain the functional activation of the gene responsible for the synthesis of the protein to guarantee its functionality, more than its presence. Moreover, from the gene expression profile, additional information that specifically correlates the expression of some genes to the response to individual therapies can be obtained; for example, in HER2-positive patients, the high expression of IGF1R correlates with resistance to Herceptin, as well as the hyperexpression of CCNE1; instead, ER+ patients with high PDGFRA expression are resistant to tamoxifen treatment [14,19,20]. The use of gene profiling tests offers the opportunity for a more adequate risk stratification, an improvement of the therapeutic planning and the clinical outcome, avoiding what happens today, with the standard clinical-pathological criteria, namely, the undertreatment of approximately 20% of women with grade 1 breast cancer and overtreatment of approximately 15% of women with grade 3 breast cancer
Methods
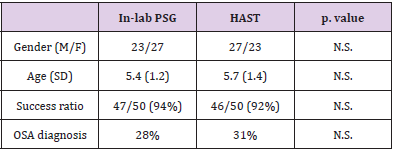
This retrospective study aimed to observe the difference in the receptor state obtained from the genome compared to that obtained with traditional immunohistochemistry. Analyzing biological material from formalin samples from 1994/1995 from two groups of selected patients with breast cancer, one consisting of patients still alive and the other of patients with a survival of less than 4 years. Survival data of a group of patients with a long follow-up period (RTUP) suffering from breast cancer diagnosed in the twoyear period 1994/1995 were obtained with the authorization of the Ethics Committee of Umbria (CEAS). The group, consisting of 55 patients, was then divided into 2 numerically balanced subgroups: the first of 30 patients staged alive on 31/12/2013 with survival of >20 years and the second of 25 patients staged in death with lower survival 4 years after diagnosis (Table 1). The relative tissue samples of the respective patients fixed in formalin and included in paraffin (FFPE) were subsequently collected and stored in the histoteca of the SC of Anatomy and Pathological Histology of the Hospital of Perugia. Twenty-two patients who had the respective tissue sample unsuitable for lack of biological material in the original inclusion block were excluded. The remaining samples, for a total of 33 cases, were all characterized again from a biopathological point of view through a dedicated immunohistochemical panel: ER, PgR, Ki67, HER2 and subjected to microscopic evaluation by a pathologist who was thus able to identify two groups of carcinomas: luminal and nonlluminal (according to the San Gallen criteria 2011 and following).
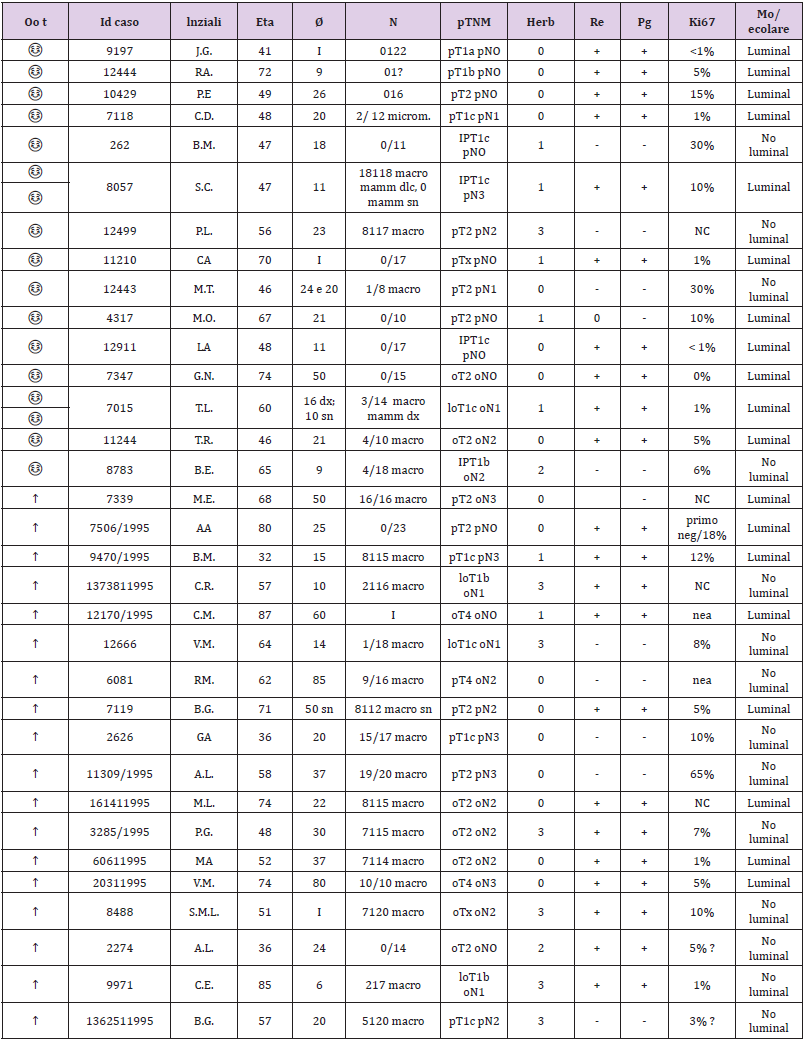
Table 1: The two-subgroup division of the 55 patients.
The recharacterization provided for the preparation of 1 slide for hematoxylin and 4 slides with blank sections necessary for the immunohistochemical panel for each sample. The pathologist made the diagnosis based on current guidelines and verified that in each section stained with the hematoxylin of each patient there were at least 30% of the neoplastic cells out of the total. This percentage figure is necessary to maximize the extractive yield of total RNA. In practice, for samples with a number of neoplastic cells ≥ 30% of the total, it was not necessary to proceed with macrodissection. In the first phase of the experimentation, so with the 33 starting samples, the enrichment of the sample was not necessary, but 2/4 sections of the FFPE fabric of 10 μm thickness were cut in sequence on microtome and placed in 1.5 ml safe look tubes ready to be extracted. Total RNA, after appropriate quantitative and qualitative evaluation, was used for the preparation of cDNA for sequencing purposes. The library used is Illumina’s TruSeq Total RNA. In the second phase of the experimentation, 2 pilot samples were used to validate the panel of genes obtained with the first phase; they were homogeneous both as biopathological characteristics of the lesion (early breast cancer; tumor diameter; lymph node status; state of HER2 and so on) and as the age of the patients. In the end, for each patient, we obtained 2 tubes of tumor tissue and 2 tubes of healthy tissue ready to be extracted. The extraction kit used to obtain total RNA was the same as that used in phase I of the trial. Total RNA, after appropriate quantitative and qualitative evaluation, was used for the preparation of cDNA for sequencing purposes. The library used was Illumina’s TruSeq RNA Access, suitable for processing RNA extracted from paraffin samples. Total RNA was extracted from sections of paraffin tissue using the “Tissue Preparation Reagents” kit – Sividon Diagnostic, from the Pathological Anatomy Section of the Santa Maria Della Misericordia Hospital. The chosen extraction method was previously “validated” on 2 samples with characteristics of “age of the sample”, “type of tissue” and “origin” identical to the samples to be used for this work. Validation test of the extraction method suitable for the preparation of the “TruSeq RNA Sample Prep” library from RNA for sequencing.
For the deconvolution of the data from the HiSeq sequencer, to process them, the following steps were carried out:
a. Demultiplexing: phase necessary to attribute to every single sample its respective data (1 sample = 1 fastq file).
b. Fastqc: phase in which the quality control of the sequencing is carried out through the use of the “fastq file for reads quality” tool.
c. Trimming: delicate but essential phase of the deconvolution process because it eliminates the reads and low-quality fragments from the Fast file.
d. Mapping: important phase of the process because for each sample a same file (also called bam) is built which specifies how the reads align on the reference genome.
e. Count table assembly: a final phase that includes all the information from the same file of the analyzed samples and related “count reads” in a single table.
Based on the reference human genome, the following are the data of the number of reads that align: Human Genome Assembly (GRCh37/hg19). These data are used as a criterion for deciding which samples will be analysed.
Samples with a low quantity of exons were excluded: a low number of reads mapped to exonic regions <12%.
Two R/Bioconductor 3.2.2 packages were used for statistical analysis:
1. DESEq2 (Love MI, Huber W, Anders S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol. 2014; 15 (12): 550.)
2. edgeR (Robinson MD, McCarthy DJ, and Smyth GK (2010). “edgeR: a Bioconductor package for differential expression analysis of digital gene expression data.” Bioinformatics, 26, pp. -1)
The edgeR package was used on the data analyzed with DESeq2 to confirm the results obtained with this calculation algorithm. Both are based on the negative binomial distribution but use different correction tests. The use of the two packages allows obtaining a list of genes that for both calculation algorithms are differentially expressed between the samples.
The statistical significance that will be used in the two packages is shown below:
1. DESeq2: P-value <0.05; P adjusted value <0.1
2. edgeR: P-value <0.05; False Discovery Rate <0.1.
An analysis of the network of genes significantly differentially expressed in the two groups was also conducted using GeneMania (http://www.genemania.org). This network was built based on the interactions between the genes, as reported by results published in the literature. Features such as gene coexpression, proteinprotein interactions, physical interactions between genes and other functions as described in the studies indicated in the table compared to the network figure are evaluated. Although paraffin introduced non negligible “background noise” in the analysis, the number of reads obtained was satisfactory to conduct a differential expression analysis using a “Read Counter” approach. It is possible to evaluate the formation of 2 small subclusters, and the analysis of differential expression with this dataset allows us to differentiate 25 genes differentially expressed between the 2 groups. The hierarchical cluster of significant genes shows a homogeneous trend among the 2 groups except for 2 luminal samples, which, although clustered with nonluminal samples, are very close to samples of the same stage (sample 7506 luminal staged due to death is very close to sample 2626 nonluminal staged due to death; sample 10429 luminal staged in life is close to sample 262 nonluminal staged in life). The IFIT3 and MX1 genes, among the 25 differentially expressed genes, have a strong interactome with other genes not present in the list but strongly correlated with each other in biological processes.From a careful observation of the PCA luminal vs nonluminal of the samples selected in Figure 1, it is possible to observe the formation of 2 groups of samples that do not comply with the luminal/nonluminal classification provided, as shown by the PCA. Although paraffin introduced nonnegligible “background noise” in the analysis, the number of reads obtained was satisfactory to conduct a differential expression analysis using a “Read Counter” approach. It is possible to evaluate the formation of 2 small subclusters, and the analysis of differential expression with this dataset allows us to differentiate 25 genes differentially expressed between the 2 groups. The hierarchical cluster of significant genes showed a homogeneous trend among the 2 groups. Among the genes, the FGFR3 gene is highlighted: a 2012 paper may be useful in which the following is stated: “FGFR3 activation in MCF7 cells stimulated activation of the mitogenactivated protein kinase (MAPK) and phosphoinositide 3-kinase (PI3K) signaling pathways, both of which have been implicated in tamoxifen resistance in breast cancer”. It should be considered that it is 4 times less expressed in “luminal” cases than in “nonluminal” cases.
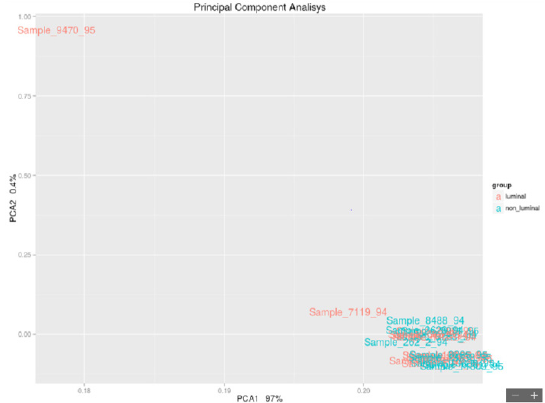
Figure 1: Luminal vs Non-Luminal.
Results
From the comparison of the results obtained with the two methods of statistical analysis, 62 significant genes emerged between living and deceased patients (Figure 2). In particular, 12 overexpressed genes emerged in live patients, and 50 overexpressed genes emerged in deceased patients. The 8 intersecting genes of the diagram are IFITM10, CEACAM7, KCNE4, OGDHL, NPY1R, SNCG, ARHGAP23, and PIEZO2. The unique DESeq 2 gene is RBFOX1. In this study, the data of the 26 samples (previous analysis) were analyzed again to highlight the genes that are differentially expressed in the luminal (life/death) and in the nonlluminal (life/ death) samples using the bioinformatic tools edgerR and DESeq2 in the R/Bioconductor environment using the following settings: DESeq2 pValue <0.05, P adjusted value 0.1; edgerR P-value 0.05, FDR 0.1. The parameters are those used in point 4.2 so that the results can be compared.
The final result is divided into some innovative cornerstones: from the 28 overexpressed genes (23 in the luminal and 5 in the nonluminal), we found that
1. DSCAM-AS1 is a specific gene of the luminal A subtype that is not present in healthy tissue or preneoplastic lesions. If this gene is present, the patient could avoid standard PBI and adjuvant therapy.
2. ER+ patients (good prognosis) died because of a high risk for the overexpression of CCND1, INST4, and GAB2.
3. ER+ patients (good prognosis) died because the overexpression of FOXJ3, SOX2, and FGFR3 (4 times less expressed in the luminal region) correlated with the failure of endocrine therapy.
4. in HERB2+ patients, we found other genes, among which PAX8-AS1 was responsible for stem cell proliferation.
5. The fusion of IFITM10 and CTSD promotes cell proliferation of the tumor and is a tumor marker.
The study highlighted a code of 33 genes that characterize breast cancer. Forty-three percent of the isolated genes were common between the 1st and 2nd sequences. Seven of our genes are found in the commercial genetic tests PAM50, Oncotype and Endopredict: four are present in the Panel Cancer targets 50 genes of the Ion AmpliSeq. The verification of the validation of the 28 genes selected from phase I in a more recent sample of women (2005) and characterized by the molecular split point (Endopredict®) allowed us to provide the study with greater consistency (Table 2).
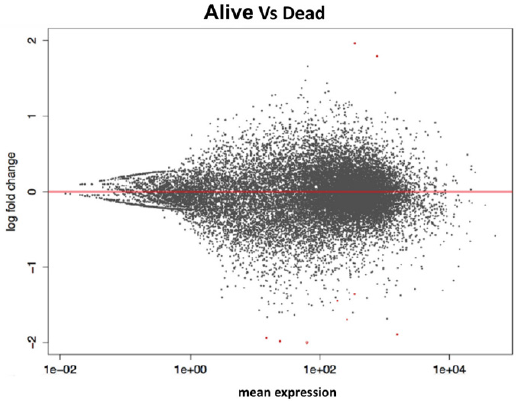
Figure 2: Life vs Dead.
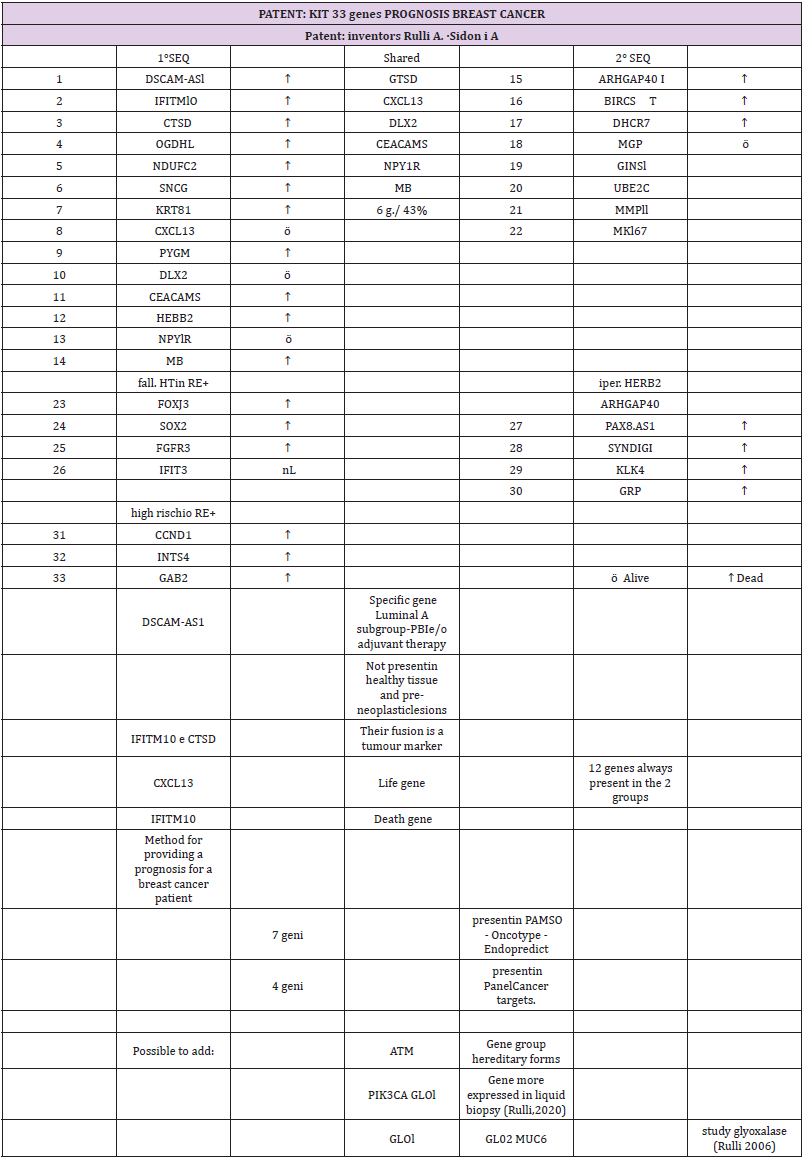
Table 2: The 28 genes characterized by the molecular split point (Endopredict®).
Conclusion
A very positive result was the ability to extract suitable RNA from 1994 samples in paraffin in good quantities and qualities to make the study possible. In this regard, experiments have been conducted to define the best protocol between RNA extraction and library preparation. The calculation of the RNA (RNA integrity number) and the concentration at Qubit allowed us to always “select” valid samples. The basic hypothesis of the study has been confirmed: the characterization of the luminal and nonluminal tumors is not real through IHC (surrogate St. Gallen), which is routinely used but must be read on a molecular basis. However, the most relevant data are represented by the 33 overexpressed genes: 28 genes (23 in the luminal and 5 in the nonluminal) and 5 genes (which confirm the premises of the study in wanting to find molecular markers capable of “personalizing” the therapy). Two of the 28 genes were always present in both groups: a CXCL13 life gene and an IFITM10 death gene. Moreover, IFITM10 and CTSD fusion promotes cell proliferation of the tumor and is a tumor marker. The DSCAM-AS1 gene is specific to the luminal A subtype. If this gene is present, the patient could avoid standard PBI and adjuvant therapy. The result obtained, which can be assumed to be transformed into a genetic panel, following validation on more recent samples (2005) and studied with Endopredict®, will help us to implement a personalization of the therapy: surgical and adjuvant. In the preoperative phase, with the core biopsy of the neoplasm, the histological diagnosis is obtained, and then the biopathological characterization and the presence of the genes described above are verified to evaluate the risk of local and/or systemic recurrence, which varies according to the molecular subtype.
The hyperexpression of CCND1, INST4 and GAB2 changes the prognosis of ER+ patients from favorable to inaustic. Our work also revealed that ER+ patients died (in which we would have expected a good prognosis) because they had overexpressed FOXJ3, SOX2, and FGFR3 genes that correlate with the failure of endocrine therapy. In the postoperative phase, targeted therapy allows a better stratification of adjuvant therapies based on the amplification of the genes that regulate, for example, resistance to tamoxifen and/ or to trastuzumab.
For More Articles: Biomedical Journal Impact Factor: https://biomedres.us